IndicatorsIt is possible to measure the pH of a solution by using a pH meter. However there are simpler ways to measure the pH. An indicator indicates the pH of a solution. An indicator changes color based of of the acidicity and basiicy of the solution. (Otherwise known as the pH) They are known as pH indicators because of this. You can not tell the pH of a solution by looking at it. There are indicators that reveal if an unknown solution is acidic neutral or basic.
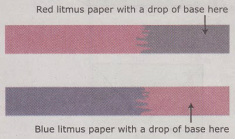
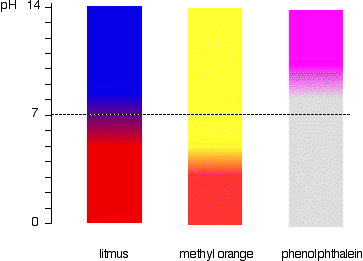
There is blue litmus paper and red litmus paper. There litmus papers reveals weather a solution is acidic or basic.
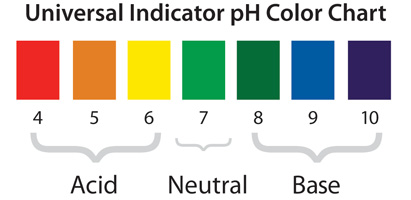
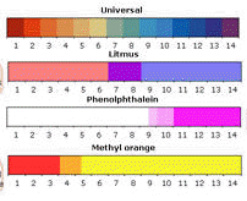
-When blue litmus paper is dipped in a acid, it turns red. -When blue litmus paper is dipper in a base, the color stays the same. -When red litmus paper is dipped in a acid, the color stays the same. -When red litmus paper is dipped in a base, it turns blue. Then there is universal indicator. Universal indicator is a solution which undergoes several color changes depending on the pH of the solution. Universal indicator is composed of several different indicators. So, with universal indicator, you are able to determine the specific pH of a solution. As shown below, a strong acid with a pH of 0-4 turns red with the universal indicator. If the solution has a pH of 5, the solution will turn orange. If the solution is a weak acid with a pH of 6, the solution will turn yellow. When the universal indicator is added to a neutral solution, then solution will turn light green. When the indicator is added to a weak base with a ph of 8, the solution will turn dark green. If the base has a pH of 9, the solution will turn blue. When the Universal indicator is added to a strong base with a pH over 10, the solution will turn purple when reacted with universal indicator.
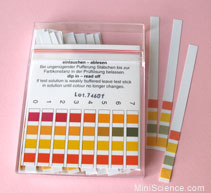
pH strips are also used to measure the pH of a solution. Click here to learn more about pH. The pH strip changes color based on the pH of the solution. pH strips are very versatile because they can determine the pH from 0 - 14. As shown in the picture to the right, once you dip the pH strip into a solution you match the colors up with the ones on the box to determine the pH across the entire scale.
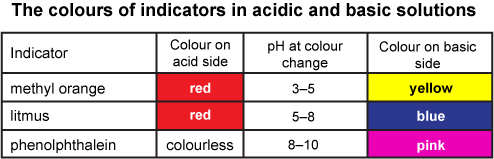
Phenophaline is commonly used in a srtong acid and strong alkali or weak acid and strong alkali titration. It will turn bright pink in a base with a pH over 8.2 and will stay clear in an acid and a neutral solution.
Methyl orange turns red at a pH lower then 3.1 and turns yellow at a pH higher then 4.4. Methyl orange is used to find out the end point of a strong acid and strong alkali or a strong acid and weak alkali titration.
To learn more about titrations, click here.
|
Where litmus, methyl orange and phenolphthalein change color in different pH solutions: Another chart showing the color changes of the indicators (universal indicator, litmus, phenolphthalein and methyl orange) depending on the pH of the solution: Picture of how universal indicator appears in solutions with a pH ranging for 1 - 14: This is a box of pH strips with a guide describing the colors for the pH: Here is a picture of a pH strip dipped in a solution with a pH in between 3 and 4: |