Conjugate acids and bases
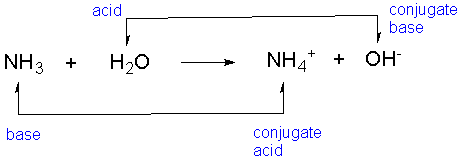
As said before, the Bronsted Lowry theory states that all acids are proton (H+) donors and bases are proton acceptors. A conjugate acid is formed when a base accepts a proton and a conjugate base is formed when an acid looses a proton.
For example : NH₃ + HCl --> NH₄⁺ + Cl⁻
In this equation NH₃ gained a proton, forming NH₄. This means that NH₃ is the base and NH₄ is the conjugate acid. In addition to this, HCl lost a proton forming Cl⁻. This means that HCl is the acid and Cl⁻ is the conjugate base.
Another example : CH₃CO₂H(aq) + H₂O(l) --> H₃O + (aq) + CH₃CO₂⁻(aq)
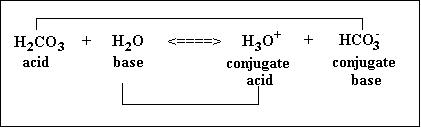
In this equation, CH₃CO₂H looses a proton (H+) and forms CH₃CO₂⁻. This means that CH₃CO₂H is an acid and CH₃CO₂ is the conjugate base. On the other hand, H₂O gains a proton and forms H₃O. This means that H₂O is the acid and H₃O is the conjugate acid.
Still don't get it?
Take this quiz
For example : NH₃ + HCl --> NH₄⁺ + Cl⁻
In this equation NH₃ gained a proton, forming NH₄. This means that NH₃ is the base and NH₄ is the conjugate acid. In addition to this, HCl lost a proton forming Cl⁻. This means that HCl is the acid and Cl⁻ is the conjugate base.
Another example : CH₃CO₂H(aq) + H₂O(l) --> H₃O + (aq) + CH₃CO₂⁻(aq)
In this equation, CH₃CO₂H looses a proton (H+) and forms CH₃CO₂⁻. This means that CH₃CO₂H is an acid and CH₃CO₂ is the conjugate base. On the other hand, H₂O gains a proton and forms H₃O. This means that H₂O is the acid and H₃O is the conjugate acid.
Still don't get it?
Take this quiz
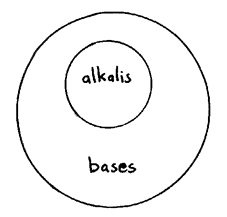
Alkali's
An alkali dissolves in water but not all bases can dissolve in water.
This also means that all alkali's are bases, and not all bases are alkali's.
This also means that all alkali's are bases, and not all bases are alkali's.