Titrations
Titrations are used to find the exact neutralizing point of a substance. They are also used to find the concentration of different substances. This titration is going to be a strong acid and strong base titration with HCl and NaOH. The products of this titration will be salt + water since we know that this is a neutralization reaction with an acid and a base. If you do not feel comfortable with neutralization reactions then click here. If I told you to use 10cm3 of 1M NaOH, through a successful titration, you would be able to tell me the molarity of HCl and the volume that was needed to execute a neutralization reaction. You would do this by averaging your results in this experiment. The results of this experiment should be 10cm3. Once you know this value, you are able to plug the number into the equation:
MA + VA = MB + VB
M = molarity
V = volume
A = acid
B = base
(1M HCl)(10cm3) = (xM NaOH)(10cm3)
Solve for x!
1M HCl * 10cm3 = 10
10/10 = 1
x = 1M
That is how you calculate the molarity of NaOH.
MA + VA = MB + VB
M = molarity
V = volume
A = acid
B = base
(1M HCl)(10cm3) = (xM NaOH)(10cm3)
Solve for x!
1M HCl * 10cm3 = 10
10/10 = 1
x = 1M
That is how you calculate the molarity of NaOH.
|
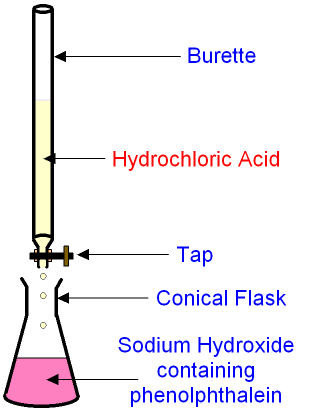
I'm going to walk you through the necessary steps that you need to do in order to do an accurate titration with HCl and NaOH.
First you must collect your materials. You will need:
Overview: This is the neutralization reaction that is happening : NaOH + HCl --> NaCl + H₂O Click here to learn about neutralization reactions Since it took 10cm3 of acid to neutralize 10cm3 of base there is no limiting reagent. |
Materials used in the titration lab:
Video of the indicator, phenolphthalein being added to the NaOH:
One drop can neutralize the solution:
|