Intro to pH, pOH, H+ and OH-
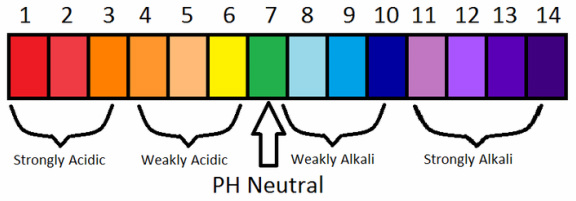
pH means potential of H+ ions. There are 14 numbers that make up the pH scale. So 0-3 would be strong acids, 4-6 would be weak acids. 7 would be neutral. 8-10 would be weak bases and 11-14 would be strong bases.
The same rules apply to pOH scale. pOH means the potential of OH- ions. There are 14 numbers that make up the pOH scale. 0-3 would be strong bases, 4-6 would be weak bases. 7 would be neutral. 8-10 would be weak acids and 11-14 would be strong acids. Capisce?
Now what you must remember is that pH + pOH = 14
Lets test this out. Lets say a liquid has a pH of 5.
14 -5 = 9 - That means that its pOH is 9.
The same rules apply to pOH scale. pOH means the potential of OH- ions. There are 14 numbers that make up the pOH scale. 0-3 would be strong bases, 4-6 would be weak bases. 7 would be neutral. 8-10 would be weak acids and 11-14 would be strong acids. Capisce?
Now what you must remember is that pH + pOH = 14
Lets test this out. Lets say a liquid has a pH of 5.
14 -5 = 9 - That means that its pOH is 9.