Intro:You will come upon acids and bases regularly in both chemistry and day to day life. Every solution is either acidic, basic or neutral. In 1887 Svante Arrhenius defined acids and bases. He found that when you put solutions into water, sometimes they would release hydrogen ions (H +). At other times, he noticed the release of hydroxide ions (OH-). He then came to the conclusion that when hydrogen ions are released, the solution must be acidic and when hydroxide ions are released, the solution is basic.
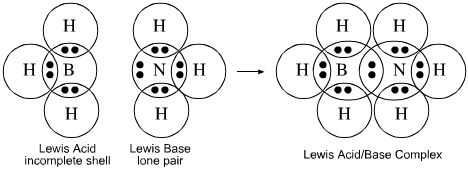
Example 1. NaOH(s) + H2O(aq) --> Na+(aq) + OH-(aq) *NaOH plus water produced OH- ions. This means that NaOH is a base. Example 2. HCl(aq) --> H+ (aq) + Cl- (aq) *HCl in water produces H+ ions. This means the HCl is an acid. The Bronsted-Lowry theory adds to the Arrhenius theory. The Bronsted-Lowry theory states that an acid is a proton doner and a base is a proton accepter. Example 1. HF + H₂O --> F⁻ + H₃O⁺* HF looses a proton and forms F⁻. This means that HF is an acid and F⁻ is a conjugate base. In addition, H₂O gains and proton and and forms H₃O⁺. This means that H₂O is a base and H₃O⁺ is the conjugate acid. The Gilbert N. Lewis introduced a new theory in 1923. The Lewis theory states that an acid can accept a pair of electrons and a base can donate a pair of electrons. For example: |
Quick Overview:
- The Arrhenius theory states thats acids are substances which produce hydrogen ions in solution. Bases are substances which produce hydroxide ions in solution.
- The Brönsted-Lowry theory defines an acid as a proton donor and a base as a proton acceptor.
- The Lewis theory states that an acid can accept a pair of electrons and a base can donate a pair of electrons.