Salts
|
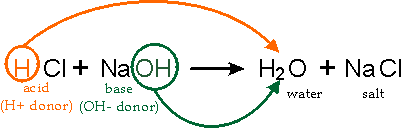
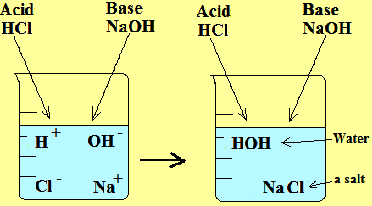
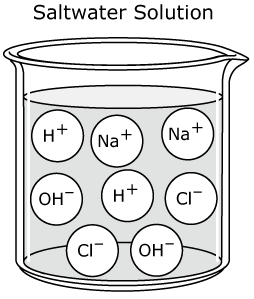
Any neutral ionic compound is a salt. Salts are made as a result of neutralization reaction. Therefore, salts are neutral. There are different families of salts that are produced. These salts are dependent upon the acid.
- If you want to make any chloride you would have to use HCL. For example: HCL + NaOH ---> NaCl + H2O Hydrochloric acid + sodium hydroxide --> sodium chloride + water - If you want to make any sulfate you would have to use H2SO4.
For example: H2SO4 + NaOH ---> Na2SO4 + H2O Sulfuric acid + sodium hydroxide --> sodium sulfate + water - If you want to make a nitrate you would have to use NO3 For example: HNO3 + NaOH ---> NaNO3 + H2O Nitric acid + sodium hydroxide --> sodium nitrate + water - If you want the metal to change you have to use a different hydroxide. For example: HCl + Mg(OH)2 --> MgCl2 + H2O Hydrochloric acid + magnesium --> magnesium chloride + water |
Feel like you know everything? |